GxP-Services Team
From calibration and qualification of complex equipment to turn-key compliance projects, our GxP Services team can provide the skilled personnel, high-accuracy measurement technology, and the appropriate documentation format for your job.
We employ more than 250 engineers, technicians and skilled workers nationwide for all tasks in the GxP-regulated environment. Through structured induction and continuous training in the complex areas of GMP compliance, we lay the foundation for the successful implementation of your measures.
Benefit from the many years of experience we have gained in carrying out a wide variety of projects.
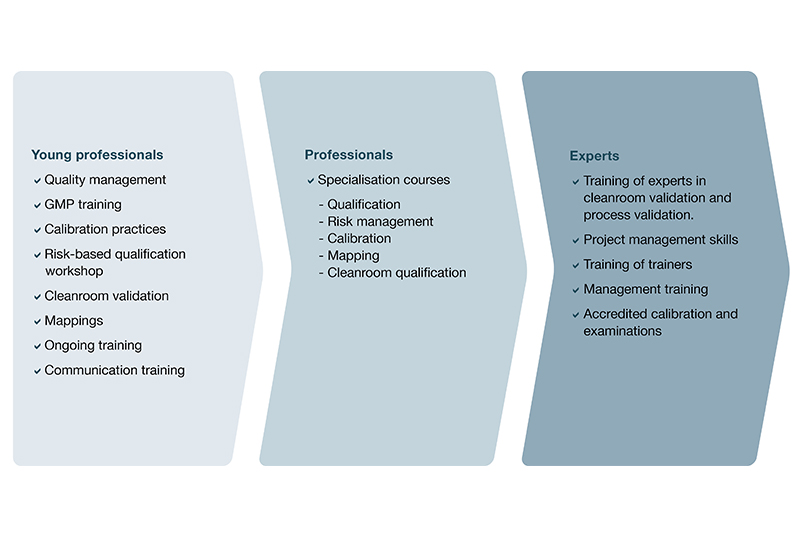
Education and training
In order to be able to implement our broad service portfolio with the highest quality at your site, we attach great importance to the training and further education of our GxP services team. We bundle specialist and expert knowledge in interdisciplinary teams - consistently geared to your needs.
Practical training
Cleanroom Training Center
At our location in Kirchzarten near Freiburg we offer our employees optimal training conditions. In our GMP training center with fully functional clean room as well as at our training facility for calibrations, our employees and seminar participants are familiarized with clean room measurements and calibrations.
Facts about the Cleanroom Training Center:
- Clean room incl. airlock on an area of 90m2
- Seminar rooms and catering can be booked
- Adjustable to ISO classes 5-8 (GMP: B-D)
- Differential pressure cascade and zone concept
- GMP-compliant, hygienic design
- Glass fronts for the spectator area
- Practice filter for clean room measurements
In our video you can learn more about the cleanroom training center as well as the preparation of our employees for their work on your site.
 Opens video in overlayThe setting of Youtube cookies is blocked until an active click on the play button is made. With this click, you give your consent for Youtube to set cookies on the terminal device you are using. You can find more information on our data protection page.
Opens video in overlayThe setting of Youtube cookies is blocked until an active click on the play button is made. With this click, you give your consent for Youtube to set cookies on the terminal device you are using. You can find more information on our data protection page.
Training facility for calibrations
Our training facility for calibrations enables practical training in the calibration of various test equipment in the following measurands:
- Temperature
- Humidity
- Speed
- Flow rate/level
- Conductivity
Extensive measuring equipment
In order to fulfil your metrological tasks, our employees have access to our large measuring equipment park. With more than 4,500 references and measuring systems calibrated according to DAkkS specifications, we cover almost all pharmaceutically relevant measured quantities.
Insight into our equipment park:
- 17 x reference sets for clean room measurements (includes particle counter, aerosol generator, dilution stage)
- 12 x reference sets for the calibration of the measurand pressure
- 1.400 x temperature and humidity loggers
- 76 x Logger for the validation of sterilization processes
- Numerous aids such as temperature baths, blocks and humidity generators


Field-tested documentation
You receive individual documentation formats according to your wishes:
- Our tried-and-tested GxP-compliant documentation format or, optionally, an individual creation of the documents - integrated into your document management system
- Prompt preparation of documentation at your site with review discussions
- We provide you with a cost efficient solution with a documentation creation that is adapted to your individual requirements
Our areas of application

Calibration
Secure measurement results - increase quality

Test equipment management with PRIMAS
Holistic test equipment management solution for compliance with standards and directives

Qualification
Qualification & Support for GMP Compliance

Validation
Ensures process stability and robustness